Global regulatory information service Orca1
"Orca1" is an information service that utilizes AI analysis to aggregate and analyze the latest regulations and inspection data from around the world, streamlining compliance operations in the life sciences (pharmaceuticals and medical devices) industry.
Overview
In the global expansion of pharmaceuticals, it is essential to stay informed about the latest regulatory information issued by the FDA and PMDA, official compendiums (USP/CFR/ISO), and frequently occurring inspection data. However, reading through the vast amount of documents that are updated daily and analyzing their impact on a company's SOPs (Standard Operating Procedures) required a tremendous amount of time and expertise.
"Orca1" uses the latest natural language processing AI to directly analyze real-time information released by regulatory authorities worldwide. The combination of B-EN-G's deep understanding of the Japanese pharmaceutical industry and the world's most advanced regulatory data analysis AI will elevate your company's compliance strategy to the next level.
function
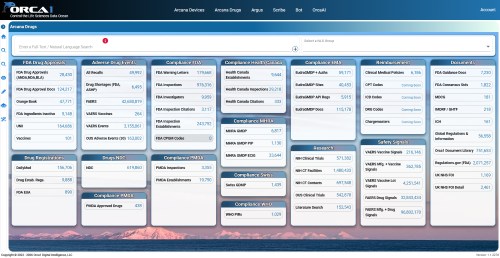
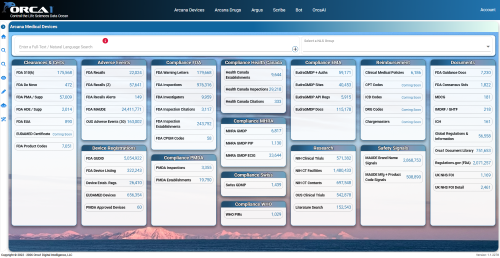
It features a variety of functions that allow you to intuitively utilize the vast amount of data collected from regulatory authorities around the world.
-
Automated generation of compliance reports by location
AI analyzes inspection history, findings, and risk scores for specific locations and manufacturing plants. It automatically generates "location evaluation reports," which previously took a considerable amount of time, in just a few minutes, accelerating supply chain selection and due diligence. - Drug application and safety information (Drug)
Comprehensive access to important data related to the drug lifecycle, such as drug application information, the Orange Book, adverse event information, and signaling information, is available. - Compliance and Inspection Information
In addition to the latest inspection data, you can check the FI score (Inspection Risk Index), whether or not there are warning letters, and the specific classification of findings. - Official citations and regulations
It allows for integrated searches with detailed regulatory references, including USP (United States Compendium) chapter numbers and UFR/CFR/ISO information. - Market and Development Trends
You can monitor over 140 draft guidelines currently open for public comment, and get real-time insights into the progress of global clinical trials (such as trials for injectable drugs). - Robust security and a flexible environment
We comply with EU and US data protection laws and strictly adhere to a non-learning policy for customer data. Deployment within a dedicated cloud environment is also possible, depending on your security requirements.
Benefits
It dramatically reduces the time and effort required for information gathering, supporting strategic decision-making.
- Dramatically speeding up SOP revision analysis
Once the draft guidelines are approved, the AI instantly performs an initial survey to determine "what needs to be modified in our company's SOPs." - Change the survey period from "six months to a few minutes."
This system reduces the initial investigation process, which traditionally took six months to a year, to just a few minutes, helping to optimize resources in manufacturing and quality control departments. - Visualizing risks and ensuring effective responses
By utilizing real-time inspection data and the latest draft information, you can visualize the risks associated with inspection responses and build a confident compliance system.
B-EN-G implementation support
As the official distributor of Orca1 in Japan, B-EN-G provides strong support to enterprise companies.
- Trusted support from official partners
As the official distributor in Japan, we provide meticulous service tailored to Japanese business practices and unique regulatory environments. - Enterprise optimization support
We support a smooth launch, from implementation to operation, in accordance with the complex internal regulations and stringent security standards unique to major pharmaceutical companies. - Providing practical application scenarios
We go beyond simply providing tools; we offer concrete advice to help you achieve results in practice, such as creating analysis scenarios for inspection data and best practices for SOP revision analysis.