Creating the future of healthcare together
Licensors' thoughts on the future of life sciences
Vol.3
Batchline
In December 2025, Business Engineering Corporation (B-EN-G) decided to make a capital investment in Thailand-based Batchline. Behind this decision is a strong commitment to provide customers in Japan's life sciences industry with next-generation manufacturing infrastructure they can rely on with operational peace of mind.
In pharmaceutical manufacturing, strict compliance withDI (data integrity) requirements and improving efficiency on the production floor often tend to be a trade-off. However, the MES from Batchline, whom B-EN-G selected as a strategic partner, achieves both at an exceptionally high level in a rapid to adopt solution.
Why did B-EN-G decide to invest in Batchline among numerous solutions and choose to partner with them? We spoke in detail with BatchlineGroup Executive Director David Margetts about their innovative design philosophy and the new value that the partnership between the two companies will bring to Japan's pharmaceutical market.(Titles omitted throughout the text)
【listener】
・Masakazu Haneda, President and CEO Business Engineering Corporation
・Yumiko Miyazawa, Director, Business Engineering Corporation, Deputy General Manager, Solutions Business Division
【Guest】
David Margetts
Batchline Group Executive Director/Global Specialist in Pharmaceutical Manufacturing DX and Compliance
He has over 25 years of global experience in digitalization, and quality management in the pharmaceutical industry.
He serves as an ISPE GAMP certified trainer, is a leading expert in CSV (Computerized System Validation) and applied data integrity. He has provided advisory services to global pharmaceutical companies for many years on strengthening compliance and improving efficiency through digitalization. He co-founded Factorytalk in 2004 and established Batchline in 2016 as a Factorytalk Group company, developing an innovative MES to resolve industry challenges.
Mission and Background
Masakazu Haneda(MH):David, thank you for joining us today. First, could you tell us about how Batchline was established and what challenges you are trying to solve in the pharmaceutical industry?
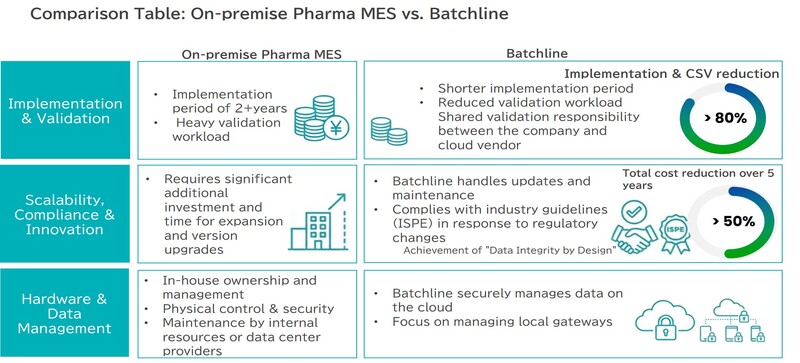
David Margetts(DM):Batchline was founded by a team of experts with decadesyears of experience at the forefront of software IT, engineering, and quality in the pharmaceutical industry. The biggest challenge we wanted to solve was how conventional MES were expensive, difficult to implement, and unfriendly to the production floor. Many pharmaceutical companies were struggling with complex system configurations and enormous validation workloads. We built a solution centered on agility that can be implemented in weeks rather than years. Our mission is to achieve remarkable cost efficiency while ensuring compliance with global regulatory standards (FDA, EMA, PICS/S), enabling pharmaceutical companies of all sizes to benefit from digitalization.
To resolve this problem:Until now, implementing pharmaceutical MES faced three high barriers. These were high investment costs in the hundreds of millions of yen, implementation difficulties spanning several years, and validation efforts involving extensive specialist work and documentation. Batchline placed agility and ease of adoption at the core of its design philosophy to overcome these weaknesses. By targeting implementation in weeks rather than years, and incorporating compliance from the design stage through a 'Quality by Design' approach, we are challenging conventional MES practices.
Significance of the Partnership and Investment with B-EN-G

MH:What strategic significance does the partnership with B-EN-G and this investment decision hold for Batchline?
DM:B-EN-G is a partner with extremely deep expertise and earned trust in Japan's life sciences industry. This investment serves as a strong endorsement that our technology meets Japan's rigorous quality standards.
DM:By combining Batchline's modern and intuitive cloud technology with B-EN-G's implementation and support capabilities specialized for pharmaceutical manufacturing cultivated over many years, we can provide Japanese customers with the highest level of compliance and peace of mind simultaneously. This is a significant first step in disseminating next-generation pharmaceutical manufacturing solutions from Asia to the world.
The Core of the Solution: Data Integrity and Compliance
Yumiko Miyazawa(YM):In pharmaceutical manufacturing, data integrity is an unavoidable topic. Specifically, how does Batchline's MES achieve both regulatory compliance (FDA 21 CFR Part 11, EMA, ISO, etc.) and on-site convenience?
DM:We consider DI (Data Integrity) not just as a checklist item, but as part of systems design and development philosophy itself. Batchline is built around system features that consider the principles of ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate + Complete, Consistent, Enduring, Available), so our customers can ensure ALCOA+ by design, and simply through use of the solution.
Batchline's MES is fully compliant with FDA 21 CFR Part 11 and EU GMP Annex 11, with robust audit trails, electronic signatures, and access control as standard features.
One of the greatest benefits after ensuring robust DI is the realization of Review by Exception. Rather than humans visually checking records and masses of documents for all processes, the system instantly extracts only deviations (exceptions) to dramatically reduce the burden on quality assurance personnel while accelerating the speed to release decisions.
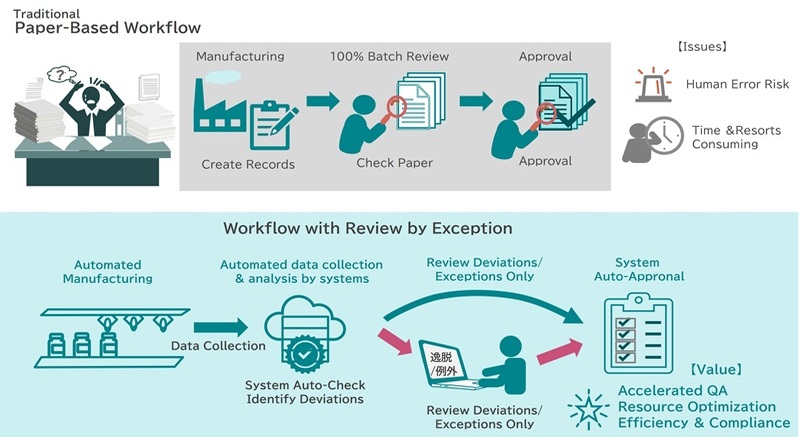
DM:Furthermore, with our mission to service all regulated manufactures, we thoroughly focused on the ease of going paperless to ensure shop-floor convenience is not compromised. For example, with intuitive operation on mobile devices and real-time error detection functions, operators can naturally follow correct procedures even when unexpected issues occur. As a result, maintaining data and record compliance is achieved through digital checks and guidance, which dramatically reduces human time and errors.
Global Development and Localization

YM:You are expanding globally while being based in Thailand. How do you view the current state of 'manufacturing DX' in markets such as the Americas, Europe, Southeast Asia, and Japan?
DM:Manufacturing DX is accelerating globally, but the challenges differ by region. In the West, digitalization is becoming commoditized and companies look for simpler and lower cost tools but already fully understand that paper is not sustainable ; in Southeast Asia, there are gaps in implementation budgets and IT literacy, and generally the industry is slower to embrace change; and Japan is characterized by extremely high quality requirements, close fit to existing procedures and careful deployment to avoid disruption.

DM:Batchline's approach includes flexible multilingual support, cloud-native scalability, connectivity with customer existing systems, and 'provision of global standard implementation models and best practice validation. Without requiring expensive on-premise servers, configurations can be quickly adapted to the language and business practices of each region. Our strength is being able to provide the highest 'global quality' manufacturing solutions from Thailand to any location worldwide, adapted to local conditions.
Due to regulations and other factors, every company will go through a "migration phase" from paper-based or simple batch record-based systems to an MES. This will not be an easy task and will require consulting services. This is an important part of our service offering.
Future vision
YM:How do you envision the global pharmaceutical manufacturing landscape changing in 5 to 10 years through the partnership between Batchline and B-EN-G?
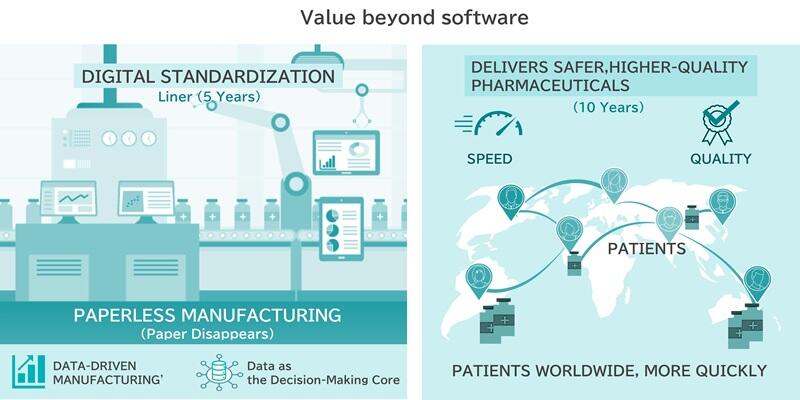
DM:Our partnership goes beyond simply providing software, both Batchline and B-EN-G are driving to a future where our software not only digitizes the current business process, but supports pharmaceutical manufacturing to move to adaptive and predictive analysis for process optimization. In 5 years, not only will 'paper' disappear from manufacturing sites, but 'data-driven manufacturing' where data becomes the core of decision-making will be standardized. In 10 years, I am confident that this platform built together with B-EN-G will become a vital infrastructure that ensures quality continuously and in real-time, visualizes the entire supply chain and ultimately contributes to delivering access to safer, higher-quality pharmaceuticals for patients worldwide.
Conclusion: Co-creation Born from 20 Years of Trust
At the end of the interview, Batchline Group Executive Director David Margetts spoke about the special relationship with B-EN-G.
B-EN-G is a company I have known and worked with for 20 years. I have always been deeply impressed by their technical capabilities, the integrity of B-EN-G’s people, and the solid track record and customer success achieved in these highly important and specialized fields.
We are very pleased to extend our Batchline core teamwith B-EN-G, and to share this exciting journey with you all.
— David Margetts
Because it is based on 20 years of critical experience, professional respect and trust, the partnership between Batchline and B-EN-G is filled with conviction that it will bring true transformation to Japan's pharmaceutical industry.
Company Profile
Batchline is part of theFactorytalk Group.20 years supplying GxP solutions and consulting across the world.
Executive Board:
・David Margetts:Group Executive Director
・David Holt:Group Executive Director
・Orchun Thakral:Head of customer success
・Neil Wetherall:Managing Director of BatchLine UK
Features:
・Recognised experts in GxP Digitalisation
・Base location : Headquarters: Bangkok, Thailand : European Office: Manchester, United Kingdom
・ISO 9001:2015 certified
・Active ISPE GAMP members & contributors to key GxP industry guidance publications
ISPE industry activities:
・”ISPE Good Practice Guide: Validation 4.0” was published in June 2025. David Margetts served as a co-lead.
https://ispe.org/publications/guidance-documents/good-practice-guide-validation-40
・"ISPE GAMP Good Practice Guide: GxP Process Control Systems 3rd Edition" was published march 2026. David margetts served as a Core member.
https://ispe.org/publications/guidance-documents/gamp-good-practice-guide-gxp-process-control-systems-3rd-edition
Company URL:https://batchline.com/company/








